 
In 1864, the English chemist John Newlands classified the sixty-two known elements into eight groups, based on their physical properties. This completes the explanation of the octet rule in this case. 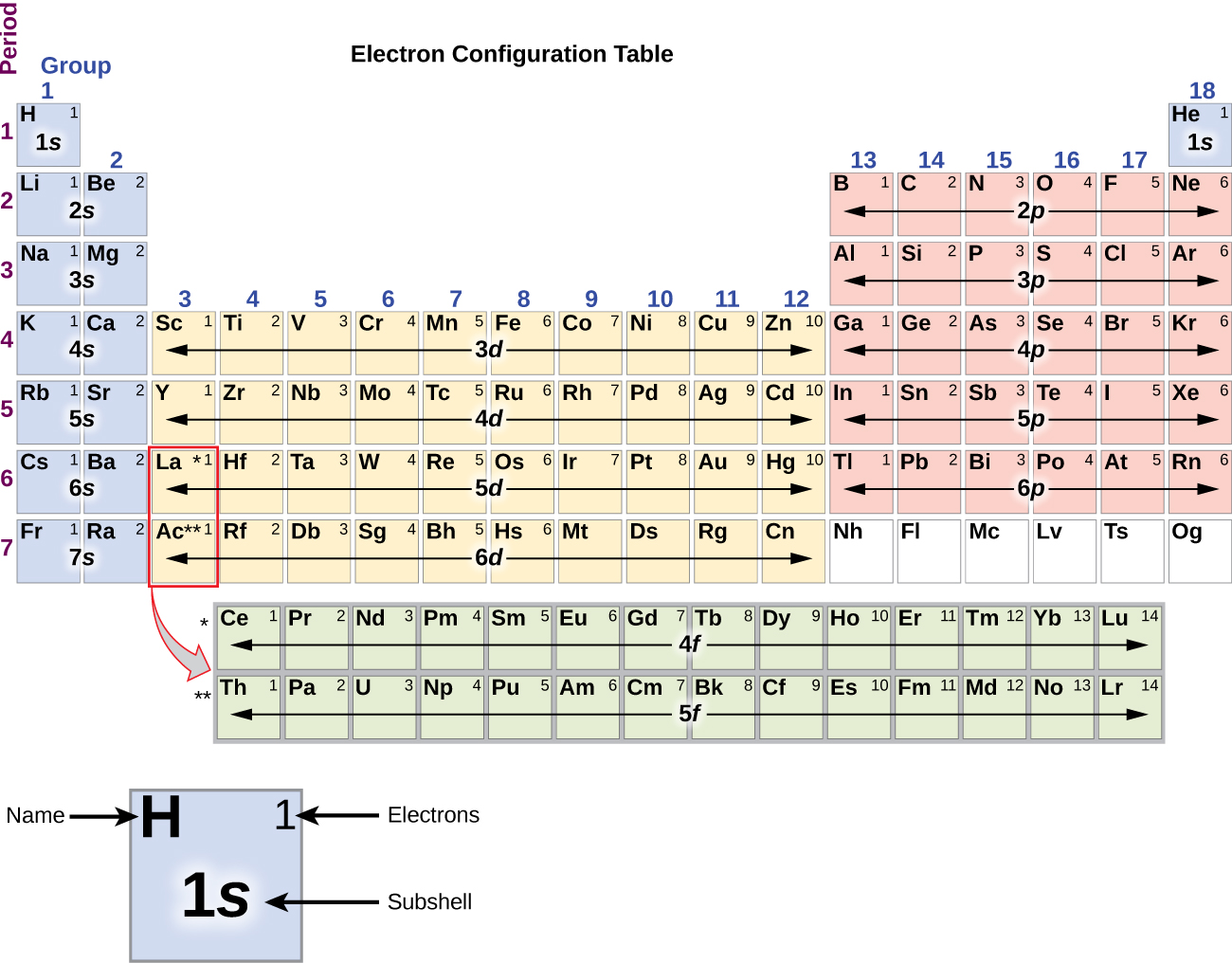
This energy is easily offset by the lattice energy of sodium chloride: −783 kJ mol −1. The energy required to transfer an electron from a sodium atom to a chlorine atom (the difference of the 1st ionization energy of sodium and the electron affinity of chlorine) is small: +495.8 − 349 = +147 kJ mol −1. Thus sodium will, in most cases, form a compound in which it has lost a single electron and have a full outer shell of eight electrons, or octet. By contrast, the second electron resides in the deeper second electron shell, and the second ionization energy required for its removal is much larger: +4562 kJ per mole. To remove this outer electron requires only the first ionization energy, which is +495.8 kJ per mole of sodium atoms, a small amount of energy. 
The result is that chlorine will very often form a compound in which it has eight electrons in its outer shell (a complete octet), as in Cl −.Ī sodium atom has a single electron in its outermost electron shell, the first and second shells again being full with two and eight electrons respectively. Adding a second electron to form a hypothetical Cl 2- would require energy, energy that cannot be recovered by the formation of a chemical bond. The first electron affinity of chlorine (the energy release when chlorine gains an electron to form Cl −) is 349 kJ per mole of chlorine atoms. Ionic bonding is common between pairs of atoms, where one of the pair is a metal of low electronegativity (such as sodium) and the second a nonmetal of high electronegativity (such as chlorine).Ī chlorine atom has seven electrons in its third and outer electron shell, the first and second shells being filled with two and eight electrons respectively. All four of these electrons are counted in both the carbon octet and the oxygen octet, so that both atoms are considered to obey the octet rule. In carbon dioxide each oxygen shares four electrons with the central carbon, two (shown in red) from the oxygen itself and two (shown in black) from the carbon. The electrons shared by the two atoms in a covalent bond are counted twice, once for each atom. The valence electrons can be counted using a Lewis electron dot diagram as shown at the right for carbon dioxide. Other rules exist for other elements, such as the duplet rule for hydrogen and helium, and the 18-electron rule for transition metals. The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens although more generally the rule is applicable for the s-block and p-block of the periodic table. 
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The bonding in carbon dioxide (CO 2): all atoms are surrounded by 8 electrons, fulfilling the octet rule. JSTOR ( October 2023) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed. Please help improve this article by adding citations to reliable sources. This article needs additional citations for verification. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
